
MyndMove™ Therapy Overview
MyndMove is a non-invasive Functional Electrical Stimulation (FES) therapy designed to improve voluntary arm and hand function in patients with upper-limb paralysis, including those recovering from stroke or spinal cord injury (SCI).
By combining electrical stimulation with active patient effort, MyndMove supports motor recovery through structured, task-specific therapy.
*MyndMove therapy is licensed in Canada for both upper and lower limb treatment applications.
How MyndMove Therapy Works
MyndMove therapy uses surface electrodes placed over targeted muscles to deliver controlled electrical stimulation while the patient actively attempts functional movements such as grasping, reaching, or pinching.
This synchronized approach—voluntary effort combined with stimulation-assisted muscle contraction—enhances motor relearning and strengthens the connection between the brain and muscles.
Therapy sessions are delivered by trained physiotherapists or occupational therapists using customizable stimulation protocols tailored to each patient’s goals and level of impairment. Meaningful improvements are often observed after approximately 20 hours of therapy, although outcomes vary by individual.
Key Features of MyndMove
Advanced Neuromodulation Technology
- Embedded algorithm-based customizable stimulation protocols
- Over 30 pre-set functional movement sequences
- 8-channel stimulator enabling activation of up to eight muscle groups
- Supports bilateral arm movement (commonly used in SCI) and complex unilateral movement (commonly used in stroke rehabilitation)
The system is designed to facilitate coordinated, task-oriented movement patterns that reflect real-world functional activities.
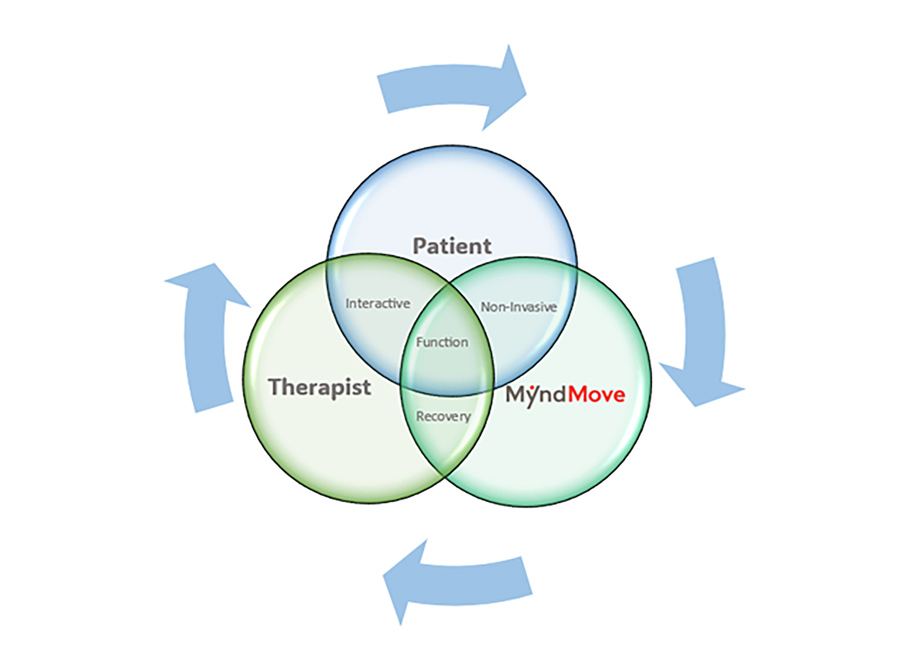
Personalized, Interactive Care
- Intuitive touchscreen interface for real-time therapy adjustments
- Protocol customization based on patient-specific rehabilitation goals
- Therapist-guided sessions focused on restoring functional movement patterns
MyndMove therapy integrates patient participation, clinician expertise, and advanced stimulation technology to promote meaningful motor recovery.
Neuroplasticity-Driven Recovery
MyndMove therapy is grounded in the principle of neuroplasticity—the nervous system’s ability to reorganize and form new functional connections.
By pairing voluntary movement attempts with precisely timed electrical stimulation, the therapy reinforces both efferent signaling (brain-to-muscle activation) and afferent feedback (muscle-to-brain sensory input).
This bidirectional activation strengthens motor pathways and supports the recovery of voluntary control in previously paralyzed muscles.
- Help reduce muscle atrophy
- Improve circulation
- Decrease muscle spasms
- Re-educate weakened muscles
MyndMove is intended for patients ranging from hemiplegia following stroke to individuals with C3–T1 spinal cord injury. Please refer to the approved indications for use in Canada and the United States.
Advanced Neuromodulation Technology
The MyndMove system is equipped with advanced embedded algorithm based, customizable stimulation protocols, enabling over 30 different pre-set movement sequences. The system’s 8-channel stimulator allows the stimulation of up to eight muscle groups in a single protocol, facilitating both bilateral arm movement (for SCI patients) and complex unilateral movement (for stroke patients).
Electrical stimulation from the device sends signals to the brain, while patients attempt movements, reinforcing a new motor pathway. Over time, this leads to lasting recovery of voluntary control in paralyzed limbs. It improves voluntary motor control and sensory function in stroke and spinal cord injury (SCI) patients, helping them perform everyday tasks.
Personalized Interactive Care
The device’s user-friendly and intuitive touchscreen interface allows therapists to tailor therapy sessions based on patient-specific goals, offering seamless customization during treatment. This technology stimulates fine motor control movements.
MyndMove’s non-invasive therapy combines patient participation, trained therapists, and a functional electrical stimulation (FES) device to restore voluntary hand and arm function in patients suffering paralysis. MyndMove promotes natural movements, offering both bilateral and unilateral stimulation protocols for patients with spinal cord injuries (SCI) and severe hemiparesis.
Neuroplasticity-Driven Recovery
MyndMove FES and NMES therapy supports recovery by not only improving movement but also preventing muscle atrophy, reducing spasms, increasing circulation, and re-educating muscles. MyndMove caters to a wide range of patients, from those with hemiplegia post-stroke to individuals with C3-T1 SCI. Please refer to the indications for use in Canada and the United States.
By stimulating neural pathways through both afferent (muscle-to-brain) and efferent (brain-to-muscle) signals, MyndMove therapy promotes neuroplasticity. Over time, this helps patients regain lasting voluntary motor control in paralyzed limbs, improving both sensory and motor functions.
Unlike some therapies, MyndMove leverages neuroplasticity by retraining the brain’s pathways through targeted electrical stimulation. Over time, patients experience lasting voluntary control of paralyzed limbs, making it a comprehensive long-term solution for improving motor function.
By combining cutting-edge technology, therapist expertise, and patient participation, MyndMove offers a powerful tool for restoring motor control in individuals with significant impairments.
MyndMove Indications for Use
Indication for Use in Canada
(licensed for upper limb and lower limb treatment)
MyndMove is an electrical stimulation device indicated for the following uses: Functional Electrical Stimulation (FES)
- Improvement of arm and hand functions and active range of motion in patients with hemiplegia due to stroke or upper limb paralysis due to C3-T1 spinal cord injury.
NeuroMuscular Electrical Stimulation (NMES) for general rehabilitation for:
- Maintenance and/or increase of range of motion.
- Prevention or retardation of disuse atrophy.
- Increase in local blood circulation.
- Reduction of muscle spasm.
- Re-education of muscles.
Indications for Use in the United States
(cleared for upper limb treatment)
MyndMove is an electrical stimulation device indicated for the following uses: Functional Electrical Stimulation (FES)
- Improvement of arm and hand functions and active range of motion in patients with hemiplegia due to stroke or upper limb paralysis due to C3-T1 spinal cord injury.
NeuroMuscular Electrical Stimulation (NMES) for general rehabilitation for:
- Maintenance and/or increase of arm and hand range of motion.
- Prevention or retardation of disuse atrophy.
- Increase in local blood circulation.
- Reduction of muscle spasm.
- Re-education of muscles.
MyndMove therapy can only be administered by Trained MyndMove Therapists who are physical and occupational therapists and also licensed or certified physical therapist assistants (PTAs) and occupational therapy assistants (OTAs) under their supervision who have completed MyndMove training, by MyndTec, on the use of the MyndMove system.
Contraindications for Use in USA and Canada
In order for patients to benefit from MyndMove, they must be medically stable, able to actively participate in and to communicate with the therapist during treatment. Conditions that preclude patients from receiving MyndMove therapy are summarized in our Contraindications for use. These include:
- Do not use the MyndMove system if the patient has a pacemaker, implanted defibrillator, or implanted metallic or electronic device. If the patient has passive metallic implants, the therapy can be delivered if the implants are located in an area other than where the electrical stimulation is to be delivered. Patients with pacemakers or an implanted electronic device should not be subjected to stimulation unless specialist medical opinion has first been obtained.
- Do not use the MyndMove system if a cancerous lesion is present or suspected on the upper extremity being treated.
- Do not use the MyndMove system on an arm if there is an unhealed wound or fracture.
- Do not use the MyndMove system over swollen, infected, or inflamed areas or skin eruptions (e.g. phlebitis, thrombophlebitis).
- Do not use the MyndMove system if the patient has cognitive impairment. Patient participation is required to deliver therapy and patients must be able to understand and follow instructions.
- Do not use the MyndMove system on an arm that has been treated with botulinum toxin in the past 6 months.
For more information on Contraindications for Use and Safety Precautions, please contact us or consult your MyndMove User Guide.
Therapist Certification
Trained Therapists Deliver Exceptional Therapy
An integral component of MyndMove therapy is the MyndMove trained therapist. Physical Therapists (PTs), Occupational Therapists (OTs), licensed or certified Physical Therapist assistants (PTAs) and Occupational Therapy assistants (OTAs) complete an intensive multi-day training course provided by MyndTec Inc. or a partner institute in order to qualify as MyndMove Therapists.
Therapists are trained to engage with patients to elicit the desired movements:
- Correctly place electrodes on the skin and optimize muscle stimulation
- Adjust stimulation levels to ensure appropriate muscle contractions
- Ensure the patient is comfortable during therapy
- Identify patient responses to MyndMove and ensure patients receive optimal therapy
Therapist Certification
Physical Therapists (PTs), Occupational Therapists (OTs), licensed or certified Physical Therapist assistants (PTAs) and Occupational Therapy assistants (OTAs) undergo a comprehensive course in the fundamental principles of MyndMove FES, device operation, protocol selection, and hands-on practice resulting in the therapist being certified for the therapeutic delivery of MyndMove.
For information on becoming a certified MyndMove Therapist, please contact us.
Clinical Evidence
The efficacy of MyndMove therapy is supported by randomized controlled trials (RCTs) in both stroke and spinal cord injury (SCI) populations. MyndMove is the culmination of years of laboratory and clinical research by Dr. Milos R. Popovic, Toronto Rehab Chair in Spinal Cord Injury Research at the Toronto Rehab Institute – University Health Network (UHN/TRI), and professor at the Institute of Biomaterials and Biomedical Engineering at the University of Toronto.1,2,3,4,5,6,7,8
Three randomized controlled studies — two in SCI and one in stroke — examined the effect of functional electrical stimulation (FES) therapy using MyndMove protocols. Patients who completed 40 one-hour sessions (3–5 times per week over 8–12 weeks) demonstrated significant and lasting recovery of voluntary arm and hand movement. Study participants represented individuals with some of the most severe motor function deficits2,5,7
.
Studies demonstrated improved arm and hand function in:
- Individuals with C3-T1 incomplete SCI2,3,5,6
- Individuals with severe hemiplegia due to stroke7,8
Outcomes were measured using:
- Self-Care Functional Independence Measure (SC-FIM™)4,7,8
- Spinal Cord Independence Measure (SCIM) Self-Care Subscore1,2,5,6
- Upper Extremity Fugl-Meyer Assessment (UE-FMA) of Motor Recovery after Stroke4,7,8
Stroke Rehabilitation
FES therapy using MyndMove protocols demonstrated lasting recovery of upper extremity function in individuals with stroke, including those with severe acute hemiplegia (2–7 weeks post-stroke) and severe chronic hemiplegia.7,8
Upper Extremity Fugl-Meyer Assessment (UE-FMA)
The UE-FMA measures volitional movement of the shoulder, elbow, forearm, wrist, and hand on a scale of 0 (minimum) to 66 (maximum). A gain of 6 points represents the Clinically Important Difference (CID).7,8
In the RCT of severe stroke, patients received either 40 hours of MyndMove therapy or 40 hours of conventional therapy (5 days per week for 12–16 weeks). The MyndMove group achieved a median UE-FMA gain of 24 points — four times the CID — while the control group showed a median gain of 0 points7,8 (P = 0.003).
Self-Care Functional Independence Measure (SC-FIM™)
The SC-FIM™ measures an individual’s ability to feed, bathe, dress (upper and lower body), groom, and toilet independently. Scores range from 6 (fully dependent) to 42 (fully independent).7,8
Before treatment, the majority of patients in both groups scored in the 6–10 range (severely dependent). After MyndMove therapy, patients shifted substantially into higher independence categories (21–30 and 31–42), while the control group showed minimal change7,8 (P = 0.005).
Additional statistically significant improvements in the MyndMove group:
- Object manipulation, palmar grip torque, and pinch grip pulling force (P < 0.05)7,8
- Barthel Index (P < 0.05)7,8
- Upper extremity Fugl-Meyer scores (P = 0.003)7,8
SCI Rehabilitation
FES therapy using MyndMove protocols demonstrated lasting recovery of upper extremity function in individuals with SCI, including those with chronic and subacute incomplete injuries at levels C3–C7.1,2,3,5,6
Spinal Cord Independence Measure (SCIM) Self-Care Subscore
The SCIM self-care subscore measures the ability to feed, bathe, dress, and groom independently, on a scale of 0 (fully dependent) to 20 (fully independent).1,2,5
Individuals with upper extremity paralysis due to SCI who underwent 40 hours of FES therapy with MyndMove combined with 40 hours of conventional occupational therapy (COT) over 8 weeks improved significantly more than those receiving 80 hours of COT alone. The difference in mean change in SCIM self-care subscores from baseline to post-treatment was statistically significant for the FES + COT group1,5 (P < 0.0001), demonstrating that MyndMove effectively increased independence and improved quality of life in individuals with tetraplegia when compared with conventional occupational therapy alone.
U.S. Department of Defense Multicentre Clinical Trial
A multicentre, single-blind, parallel-group, two-arm randomized controlled trial funded by the U.S. Department of Defense (U.S. Army Medical Research Acquisition Activity, Award W81XWH-16-1-0790) was completed comparing MyndMove FES therapy to intensive conventional therapy in adults with C4–C7 traumatic incomplete tetraplegia. Fifty-one participants were enrolled across four SCI-specialized neurorehabilitation centres in the United States and Canada, with a baseline SCIM III Self-Care subscore of ≤10. Participants received 40 one-hour sessions (3–5 per week, up to 14 weeks) of either MyndMove therapy or conventional therapy of equivalent intensity and duration.9
The trial concluded that 40 sessions of FES therapy delivered by the MyndMove stimulator were as effective as conventional therapy in producing meaningful functional improvements that persisted after therapy was completed.9
Sponsor:
- MyndTec Inc.
Collaborators:
- U.S. Army Medical Research Acquisition Activity; United States Department of Defense; Programs for Assessment of Technology in Health Research Institute; McMaster University
Trial registration: NCT03439319
Studies Referenced
- Kapadia N, Shaghayegh B, Popovic MR. Influence of Different Rehabilitation Therapy Models on Patient Outcomes: Hand Function Therapy in Individuals with Incomplete SCI. The Journal of Spinal Cord Medicine. 2014:0:0-1.
- Kapadia N, Zivanovic V, Furlan J, Craven BC, McGillivray C, Popovic MR. Functional Electrical Stimulation Therapy for Grasping in Traumatic Incomplete Spinal Cord Injury: Randomized Control Trial. Artif Organs. 2011:35(3):212-216.
- Kapadia N, Zivanovic V, Popovic MR. Restoring Voluntary Grasping Function in Individuals with Incomplete Chronic Spinal Cord Injury: Pilot Study. Topics in Spinal Cord Injury Rehabilitation. 2013:19:4-279.
- Kawashima N, Popvic MR, Zivanovic V. Effect of Intensive FES Therapy on Upper-Limb Motor Recovery After Stroke. Physiotherapy Canada. 2013:65(1):20-28.
- Popovic MR, Kapadia N, Zivanovic V, Furlan J, Craven BC, McGillivray C. Functional Electrical Stimulation Therapy of Voluntary Grasping Versus only Conventional Rehabilitation for Patients with Subacute Incomplete Tetraplegia: A Randomized Clinical Trial. Neurorehabilitation and Neural Repair. 2011:25:5-433.
- Popovic MR, Thrasher T, Adams M, Takes V, Zivanovic V, Tonack M. Functional Electrical Therapy: Retraining Grasping in Spinal Cord Injury. Spinal Cord. 2006:44-143.
- Marquez-Chin C, Bagher S, Zivanovic V, Popovic MR. Functional electrical stimulation therapy for severe hemiplegia: Randomized control trial revisited. Canadian Journal of Occupational Therapy. 2017:84(2):87-97
- Trasher T, Zivanovic V, Mcllroy W, Popovic MR. Rehabilitation of Reaching and Grasping Function in Severe Hemiplegic Patients Using Functional Electrical Stimulation Therapy. Neurorehabilitation and Neural Repair. 2008:22:706-13.

